Frogs, unlike their salamander cousins, cannot regenerate lost limbs, but a fascinating new experiment suggests it’s possible for frogs to partially regrow their missing body parts under the right conditions.
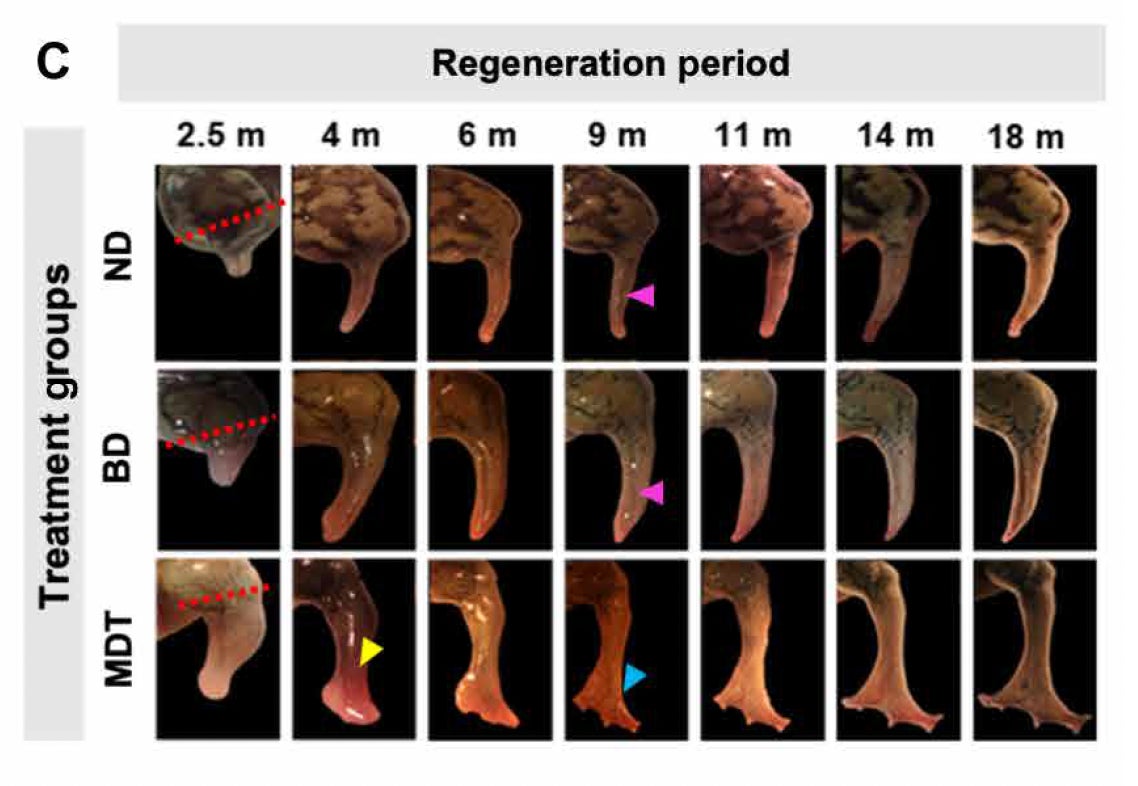
A paper published today in Science Advances describes a new treatment that made it possible for African clawed frogs (Xenopus laevis) to partially grow back their amputated hind limbs. The treatment consisted of five different drugs and a wearable device, called “BioDome.” Exposure to this chemical cocktail for just 24 hours facilitated a regenerative period that last for 18 months, according to the research, led by researchers from Tufts University and Harvard University’s Wyss Institute.
“It’s exciting to see that the drugs we selected were helping to create an almost complete limb,” Nirosha Murugan, the first author of the paper and a researcher at Tufts, said in a statement. “The fact that it required only a brief exposure to the drugs to set in motion a months-long regeneration process suggests that frogs and perhaps other animals may have dormant regenerative capabilities that can be triggered into action.”
It’s an exciting achievement, as it suggests animals otherwise incapable of spontaneous regeneration can, under certain conditions, regrow lost limbs and possibly other body parts, such as organs and tissue. This exact strategy may not work for humans, but the new paper could inform future studies in the hopes of developing something similar for mammals. That said, the new research is very preliminary, requiring more work to confirm the results and to better elucidate the mechanisms responsible for the observed regeneration.
Salamanders, starfish, crabs, and some lizards can regenerate lost body parts, but not frogs. Nor can mammals, though the human liver can regrow to full size even after 90% of it has been removed. And as scientists learned in 2019, we also have the capacity to regenerate injured or overworked cartilage in our joints. As for regrowing missing fingers or an entire leg, that’s not possible given how wound healing works in mammals; for the most part, our injuries get covered in scar tissue, which helps protect them from infection or further damage.
Michael Levin, a co-author of the new study and a biologist at Tufts, has spent more than 20 years trying to understand how batches of cells make decisions about what they’re going to build. This research is important, as the resulting insights could lead to treatments for birth defects, cancer, traumatic injuries, aging, and possibly tissue and limb regeneration.
“People have been working on limb regeneration for a long time, studying animals like salamanders which already regenerate organs,” Levin explained to me in an email. “Our approach is different from others that focus on stem cell implants or genome editing: rather than try to micromanage the process, we seek brief signals that will convince the cells to restart doing what they already did in embryonic development.”
To do this convincing, Levin and his colleagues concocted an elixir containing five drugs, which collectively boosted cell proliferation, reduced inflammation, pushed cells into repair mode, inhibited the production of collagen (which can lead to scar formation), and facilitated the growth of nerve fibers, blood vessels, and muscle.
For the experiment, the team amputated the hind limbs of 115 adult female African clawed frogs. In terms of the ethics involved, Levin said his team had “an extensive protocol, approved by a committee of veterinarians and aquatics animal experts,” and general anesthesia was used to minimize distress. “Respect for the animals is a key aspect of all our work,” he added.
The ingredients of the chemical cocktail were clearly critical to success, but so too was the bioreactor. The wearable device inhibited the usual process of closing the stump, and instead encouraged limb regeneration. Use of the BioDome helped to “mimic an amniotic-like environment which, along with the right drugs, allows the rebuilding process to proceed without the interference of scar tissue,” David Kaplan, a co-author of the study and an engineer at Tufts, said in the statement.
It took the limbs many months to regrow, but the results were dramatic. The regenerated hind legs were nearly fully functional, exhibiting similar bone structure and internal tissues (including neurons) compared to the original. The frogs even regenerated several toes, though without bones. In tests, the frogs swam through water in their typical way, and also reacted when the regenerated legs were poked.
“Our collective data suggest that early targeted interventions may shift the burden of regeneration to the limb itself, sidestepping reliance on longer-term therapeutic strategies like the use of stem cells, serialized treatments, and other means of persistent micromanagement of restorative growth and patterning,” write the scientists in their paper.
As frogs and humans are very different when it comes to their biology, I asked Levin how translatable the findings are to mammals.
“All vertebrate animals share key anatomical control machinery. If salamanders can regenerate, it’s possible for us too,” he replied. “We may need to adjust the BioDome and the cocktail, and we’re certainly not ready for human trials yet, but I think this is the path to medical regeneration, eventually. And not just for limbs—there’s nothing in that cocktail that’s limb-specific, it’s a signal that told the cells to grow what normally goes there.”
In terms of next steps, the team plans to test new cocktails in mice and then optimize the mixture as required.
The researchers speculate that a molecular pathway associated with embryonic development was activated by the treatment, but future research should investigate this further. It would also be good to see other researchers replicate these results, and dive deeper into the exact regenerative processes involved. It’s a promising start nonetheless, and an exciting new direction for future research.
More: Humans Have a ‘Salamander-Like’ Ability to Regenerate Damaged Body Parts.






