Stanford researchers think they’ve devised an effective and quick-acting way to treat difficult cases of depression, by improving on an already approved form of brain stimulation. In a new trial published this week, the researchers found that almost 80% of patients improved after going through treatment—a far higher rate than those who were given a sham placebo.
Brain stimulation has emerged as a promising avenue for depression, particularly depression that hasn’t responded to other treatments. The basic concept behind it is to use electrical impulses to balance out the erratic brain activity associated with neurological or psychiatric disorders. There are different forms of stimulation, which vary in intensity and how they interact with the body. Some require permanent implants in the brain, while others can be used noninvasively, like repetitive transcranial magnetic stimulation (rTMS). As the name suggests, rTMS relies on magnetic fields that are temporarily applied to the head.
rTMS has been approved since 2008 by the Food and Drug Administration for patients who don’t respond to at least one other treatment. But its success rate is modest, with about 14% to 30% of patients experiencing a significant remission. And while it’s safe and requires less upkeep than other forms of stimulation, a typical single course takes about six weeks to complete, which is less than ideal for patients experiencing an urgent depressive episode.
In recent years, scientists at the Stanford University School of Medicine have been trying to improve rTMS. Their technique, now dubbed the Stanford neuromodulation therapy (SNT), relies on higher-dose magnetic pulses delivered over a quicker, five-day schedule, meant to mimic about seven months of standard rTMS treatment. The treatment is also personalized to each patient, with MRI scans used beforehand to pick out the best possible locations along the brain to deliver these pulses.
“We were very interested in trying to solve psychiatric issues in an emergency setting, where we’re treating people in the time course of days. And so we figured out a way, based off of human neuroscience principles, to compress stimulation from a six-week schedule into a single day,” study author Nolan Williams, an assistant professor of psychiatry and behavioral sciences at Stanford, told Gizmodo over the phone.
Last year, Williams and his team published a small study of 21 patients who were given SNT, showing that 90% of people severely affected by their depression experienced remission—in other words, that they no longer met the criteria for an acute depressive episode. Moreover, people’s feelings of suicidal ideation went away as well. The study was open label, though, meaning that patients and doctors knew what treatment was being given. Confirming that any drug or treatment actually works requires more rigorous tests, such as a double-blinded and placebo-controlled experiment. And that’s what the team has done now, publishing the results of their new trial in the American Journal of Psychiatry.
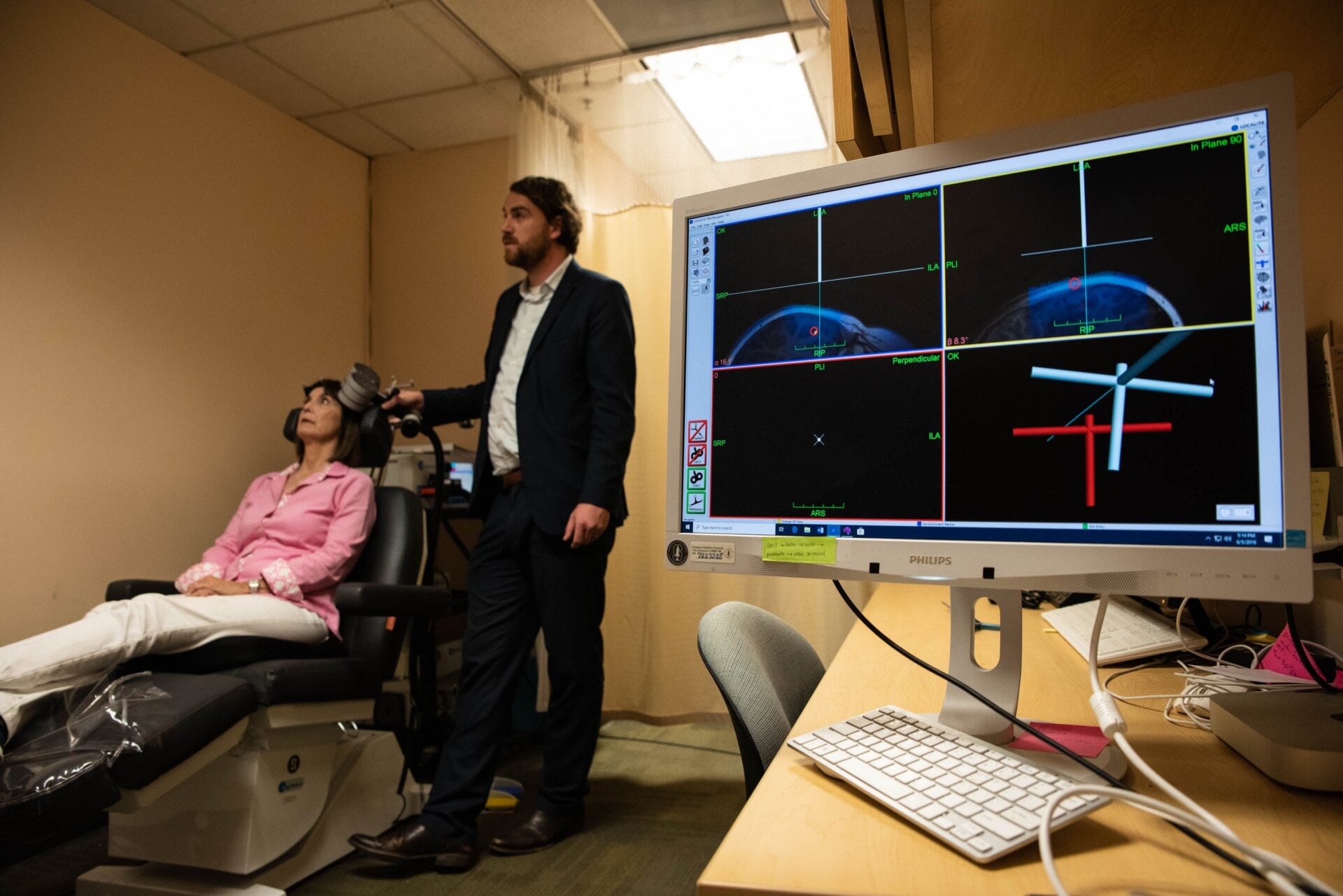
Obviously, finding a placebo for brain stimulation isn’t quite as easy as asking people to take a sugar pill in a standard drug trial. Instead, patients will go through a sham stimulation session, where the procedure is carried out to the point where patients think they’re being stimulated, but only a weak or no pulse is actually used. In this new trial, 29 participants with treatment-resistant depression were randomized to get real SNT or the sham. Afterward, they were monitored for four weeks.
This time, about 78% of patients given genuine SNT experienced remission, based on standard diagnostic tests, compared to about 13% of the sham group. There were no serious side effects, with the most common being a short-lasting headache. And when participants were asked to guess which treatment they took, neither group did better than chance, indicating that the blinding worked.
Tommy Van Brocklin, a 60-year-old Tennessee resident, wasn’t involved in the current trial. But this past September, he flew out to California to take SNT at Stanford as part of ongoing research by the team. Brocklin has struggled with depression since he was a child. And though he did experience significant relief in later life from the antidepressant Paxil, its benefits for him have steadily declined over time. This year, he began to experience constant suicidal ideation.
“By the third day, everything started to kick in. And it just got better and better the next few days,” Brocklin told Gizmodo about his experience with SNT. “I’ve been home now since about mid September and all the benefits have stayed with me, and I feel much better. I’m sleeping; I’m no longer suicidal. I just have a different outlook on the world and my life, in a positive way.”
Though the results are again based on a small sample size, an interim analysis of the trial determined that the difference in success rate was so large that it would be unneeded to continue the study any further. And the authors note that there have been other similarly sized trials testing out new therapies for treatment-resistant depression, including ketamine (a version of which is now FDA-approved).
“We were pleasantly surprised that this was more powerful, statistically, than what we anticipated. And then we felt it important to stop the trial and get the information out there as soon as we could,” Williams said. Incidentally, the trial was ended in early 2020, just before the covid-19 pandemic began in earnest and temporarily shut down the group’s lab anyway.
The sheer scale of success observed in their studies so far makes the group confident that their treatment is the real deal. Notably, it may even outperform electroconvulsive therapy, the most widely used form of brain stimulation for treatment-resistant depression, but one that requires general anesthesia and a full medical team to perform. And because SNT can be done over a week’s time, they argue, it could serve as a sort of rescue therapy for patients in crisis, allowing them to get better immediately and then transition to other maintenance treatments like medication or talk therapy (most approved treatments, including antidepressants, can take months to come into effect).
In the future, Williams doesn’t rule out the possibility that a version of SNT could be used as a maintenance treatment itself. Anecdotally, he notes that patients may show continued improvement from a five-day session for weeks to years, and that patients whose symptoms return can still benefit from additional courses.
Though SNT is a modified version of rTMS, Williams says that their technique is a truly novel application of it. The research group has obtained patents for SNT, and they are seeking FDA approval. They’ve also gotten a breakthrough designation from the FDA—a label that indicates that the FDA will expedite its review of something that shows promise in treating a serious condition.
The authors do acknowledge that more and larger trials of SNT would need to be conducted before this treatment could see wide use, which could include direct comparisons to other forms of brain stimulation. But for now, both the team and their patients are hopeful about the potential of this treatment.
“I had started to get to the point where I was getting very, very frustrated, and I thought about giving up.” Brocklin said. “So that was really one of the best things about the treatment—that it happened to help so fast.”