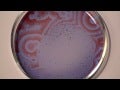
In the 1950s, Boris Belousov noticed that a certain combination of chemicals could switch off between two different reactions. He also noticed that switch could be made very, very pretty. In the early 1960s, when Anatoly Zhabotinsky noticed the same thing, and entire chemistry aesthetic was born. We’ll show you examples of the Belousov–Zhabotinsky reaction, and tell you how it works.
https://www.youtube.com/watch?v=3JAqrRnKFHo
One of the troubling things about chemistry is the fact that it seems that molecules never come to rest. Individual molecules keep trading off atoms and electrons, and appropriating different properties, even while the chemicals are, overall, in an equilibrium with each other. This lends a febrile quality to the science that’s troubling, but it can make for some very cool reactions. One such reaction was discovered by Boris Belousov sometime in the 1950s. He mixed together a group of liquids that included cerium (IV) sulfate. The combination of chemicals reacts to slight agitations. When someone put stress on the surface, it caused the cerium (IV) ion to switch to a cerium (III) ion, which slightly changed the color of the solution. It also changed the composition of the solution. That new composition turned the cerium (III) back into cerium (IV) again. The color changed back along with it. The reaction spread out from these points of agitation, and the oscillations of the reaction caused incredible, shifting patterns to form.
https://www.youtube.com/watch?v=qm_UwbPmT9k
In the early 1960s, Anatoly Zhabotinsky noticed the same phenomenon. When they presented the Belousov-Zhabotinsky, or BZ reaction, research into similar reactions began. BZ reactions are the name given to a group of oscillating reactions that treat us to a light show. Because they deal with actions and reactions spreading from certain points, they’ve also been yielded data into how some types of chemical reactions move and disperse.
And, they are always cool to look at.
Top Image: Armin Kübelbeck
Via Anthony Hall