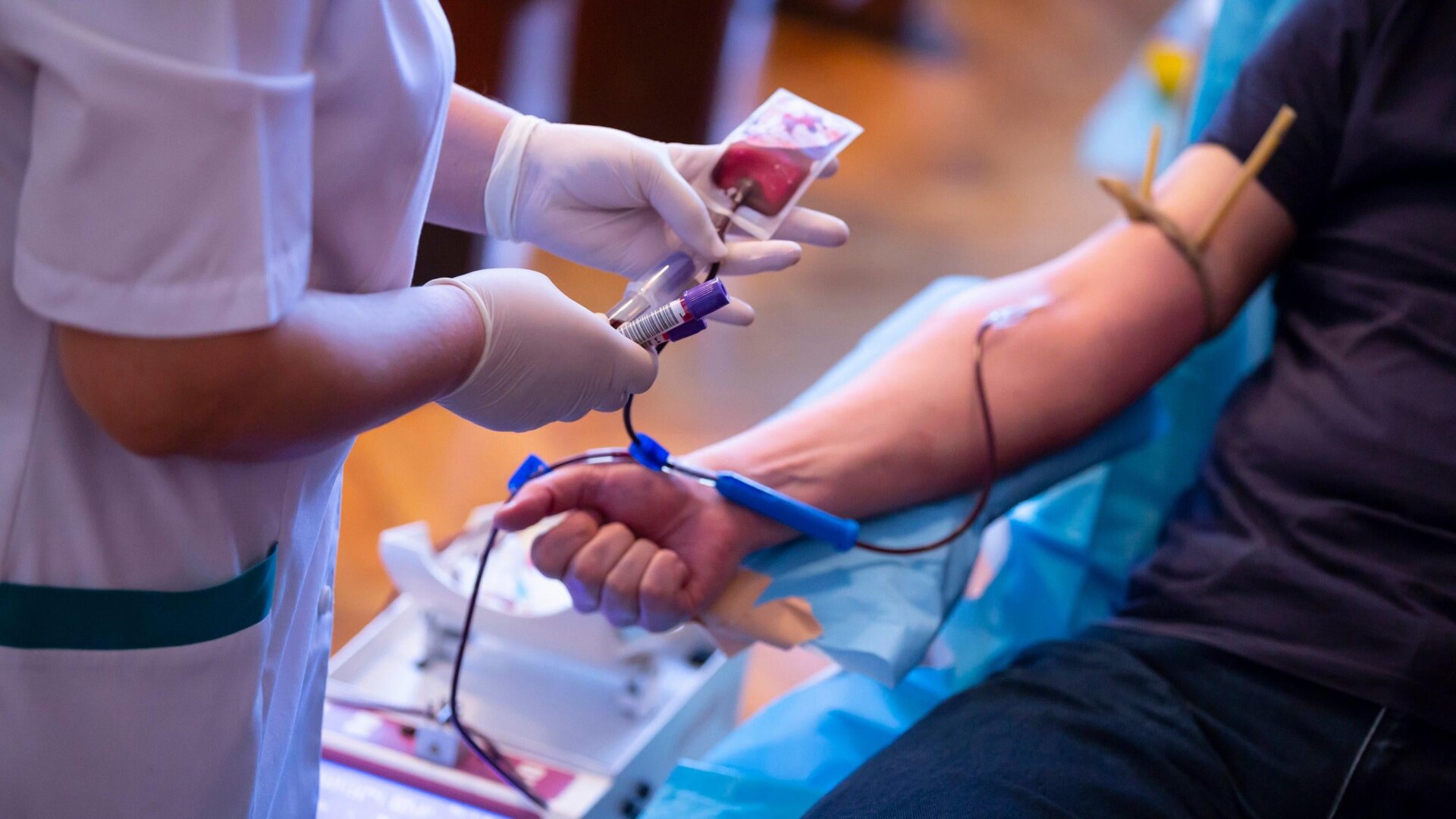
The Food and Drug Administration is about to lift a blanket restriction on gay and bisexual men donating blood, according to a report out Wednesday in the Wall Street Journal. The agency is reportedly planning to adopt a more individualized criteria for its screening process, one that will allow currently monogamous men who have sex with men to donate regularly.
For more than three decades, the FDA has set harsh restrictions on gay and bisexual men donating blood and tissue. The original rationale for the decision was that these individuals had a higher risk of contracting HIV, which could then be transmitted through the donation supply. But many experts and LGBT advocates have criticized the practice, especially as medical technology has advanced. Current tests are now able to detect HIV in donated blood more accurately and from people only recently infected, for instance.
The FDA has slowly changed its policies over time. In 2015, it removed its lifetime ban but still only allowed gay and bisexual men to donate if they hadn’t had sex with men in the past year. In 2020, the agency shortened the time period needed for abstinence to 90 days, partly as a response to the shrinking blood supply fueled by the covid-19 pandemic. But many advocates and health organizations like the American Medical Association have continued to push the FDA to dismiss these restrictions in general. They’ve argued that the screening guidelines should explicitly focus on people’s behavioral risk factors, not their sexual orientation. And it now appears that the FDA has finally listened to these recommendations.
The WSJ reports, citing anonymous sources within the agency, that the FDA is expected to drop strictly time-related restrictions for gay and bisexual men. Potential donors will instead be screened by their recent sexual history. People who haven’t had a new sex partner in the last three months will likely be able to donate with no issue, regardless of their orientation (assuming no other reasons for deferral). People who have had a recent new partner will then be asked if they’ve had anal sex during that time period and will likely be asked to wait for at least three months if they answer yes.
Anal sex and sex with multiple partners, especially if unprotected, are known to be greater risk factors for transmitting and contracting sexually transmitted infections like HIV. And the three-month waiting period is thought to be long enough for people to find out if they have caught HIV from a recent exposure, according to an FDA official cited by the WSJ. The changes envisioned by the FDA also seem to be backed by a recent FDA-funded study that tested out the new approach and involved around 1,600 gay and bisexual men.
“I’m thrilled to see this step forward,” North Carolina Health and Human Services Secretary Kody Kinsley, who has pushed the FDA to change its policy, told the WSJ. “It’s long past time for us to protect the blood supply based off what people do and not who people are.”
The FDA is reportedly still finalizing its new guidelines but is expected to roll out the changes sometime in the next few months.