The Department of Justice has brought charges against the president of a Silicon Valley-based pharmaceutical firm, Arrayit, alleging a scheme to profiteer off the coronavirus pandemic with a shoddy covid-19 test it tried to bundle with an expensive, unrelated allergen finger stick test. The DOJ said the charges are its first securities fraud case brought in relation to the pandemic, though elements of the alleged wrongdoing call to mind the Theranos debacle.
Per the Washington Post, prosecutors allege that Mark Schena committed securities fraud and conspiracy to commit health-care fraud, charges that potentially carry a combined sentence of up to 30 years in prison and fines of over $5 million. A criminal complaint filed with the U.S. District Court of the Northern District of California and signed by U.S. Postal Service inspector Anna Hallstrom claims that Schena used “kickbacks and bribes to recruiters and doctors” to bill Medicare providers and private insurers for its “expensive and medically unnecessary” allergen tests, as well as made false claims about its coronavirus testing capabilities to pump Arrayit stock. Schena also allegedly sent investors massively inflated figures on the financial health of the company in order to “justify hundreds of millions of dollars in production.”
Arrayit CLIA clinical team provides allergy testing finger stick and blood card collection reminder to all of our medical clinic and retail customers. Please make sure to fill all four card circles completely to accelerate test processing and reporting TY https://t.co/To67BQtJJq pic.twitter.com/G4ErgnWOWP
— Arrayit Corporation (@arrayit) July 31, 2018
The complaint alleges that in March 2020, Schena began emailing clinics saying that Arrayit had developed a test for the novel coronavirus based on “advanced Silicon Valley technology and finger stick blood collection,” sparking increases in its stock price. It goes on to say that “In reality, Arrayit had not developed, validated, or produced a test at that time,” and the Food and Drug Administration informed the company in April that the test it did eventually produce fell short of standards necessary to secure an emergency use authorization.
Arrayit also launched an extensive online promotional campaign on social networks like Facebook to promote the bundling of the covid-19 test with the allergen one, according to the complaint, falsely claiming that the nation’s top medical officials had endorsed using finger pinprick tests to separate symptoms of the coronavirus from those of allergies. In March, before the FDA declined to approve Arrayit’s test, the value of the company’s stock doubled from two cents to four cents. (According to previous reporting, the Securities and Exchange Commission halted trading in Arrayit stock for two weeks on April 13, saying the company hadn’t filed financial reports since 2015.)
In total, prosecutors say that Schena and others affiliated with the company submitted nearly $69 million in false and fraudulent claims, the San Francisco Chronicle reported. Of that, some $5.9 million was billed to Medicare. Schena wasn’t very lucky in collecting, however, allegedly netting over $2 million from private insurers and around $290,000 from Medicare.
The coronavirus test never actually made it to market, despite Schena allegedly claiming that bundling it with the allergen tests would be as easy as “a pastry chef” moving from “strawberry pies” to “rhubarb and strawberry pies.” The complaint says that after lying to investors about demand for the coronavirus tests, Schena never actually told them of the failure to reach “viability or availability.”
Evidence of Arrayit’s promotional campaigns remain online; a Twitter account associated with the company claimed in 2019 their allergen test was “endorsed by top television and Internet celebrity” Jake Paul, complete with a photo of him jewelry-clad and grasping another YouTuber’s buttocks. (Paul has since moved on to, err, other endeavors.)
Arrayit Pinner USA blockbuster consumer lifestyle tests feature doctor-prescribed finger stick microarrays to fine tune your diet for peak performance. Pinnertest is endorsed by top television and Internet celebrity and 12.5m Instagram sensation Jake Paul https://t.co/q9KjuPsdKA pic.twitter.com/yc4igdGR4u
— Arrayit Corporation (@arrayit) August 15, 2019
Theranos, another company that claimed to have developed next-generation finger pinprick blood tests, famously imploded after it was revealed that their blood testing machines didn’t work and the company was diluting samples to keep up the appearance of a functional company. Theranos, however, achieved a peak valuation of $10 billion on some $700 million in funding. Founder Elizabeth Holmes and former president Ramesh “Sunny” Balwani have managed to have some charges dismissed but are still facing nine counts of wire fraud.
Arrayit actually referred to Theranos in a 2015 Facebook post, claiming its own technology was far better.
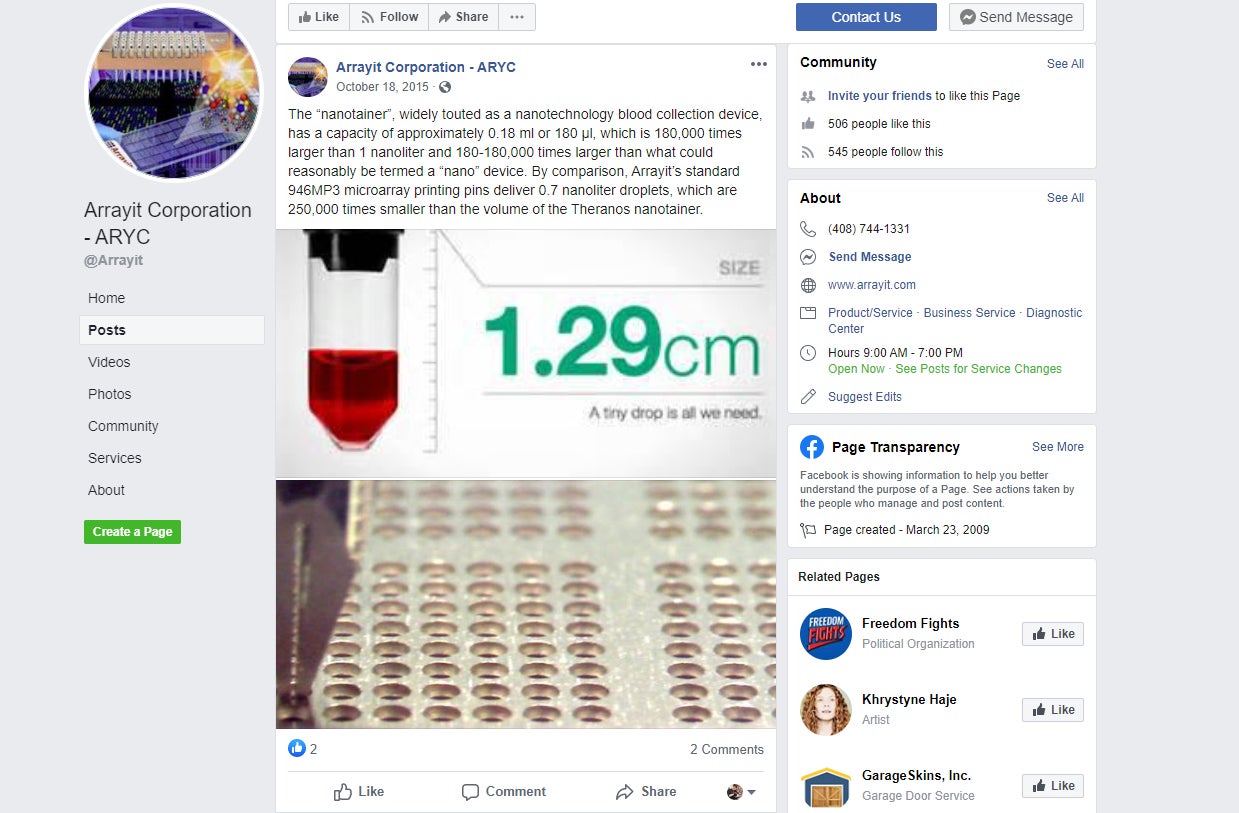
The Post said several attempts to reach Arrayit or Schena for comment were “unsuccessful.”
“Investigating COVID-19 fraud scams billed to federal health programs—such as those charged here—are a top priority for our agency,” Steven Ryan, the special agent in charge at the Department of Health and Human Services inspector general’s office, told the Mercury News in a statement. “The ongoing public health crisis has spawned a rash of fraudulent schemes; therefore, we will continue working with law enforcement partners to protect beneficiaries, programs and taxpayers.”
As of Tuesday evening, a tracker on the Johns Hopkins University School of Medicine website listed nearly two million confirmed cases of the coronavirus in the U.S., with nearly 112,000 deaths. The tracker listed over 7.2 million cases confirmed globally, resulting in over 411,000 deaths.






