The dream of nanotechnology includes unbelievably tiny electronic devices — including medical devices that could work at the microscopic level. But how do you assemble nanoparticles into larger systems, like nanocrystals? You can’t exactly use a pair of tweezers.
Instead, researchers create a solution of dissolved nanoparticles, which self-assemble into crystal structures. These nanocrystals –- which have a uniform structure and properties that can vary depending on their size –- may have a variety of applications. But to customize them, researchers need to be able to control their growth. And a good first step is figuring out how they assemble in the first place.
As the researchers write in their paper, published in the journal Science, “Understanding the mechanism of…nanocrystal growth using nanoparticles as building blocks provides a link between the world of single molecules and hierarchical nanostructures, and paves the way to rational design of nanomaterials with controlled properties.”
How can you observe a nanocrystal as it grows? While any traditional optical microscope would fail miserably if it tried to focus at this scale, there are other imaging methods, such as transmission electron microscopy, or TEM. In TEM, the device sends a beam of electrons through an object and measures how the electrons interact with the sample. Then, an image of these interactions can be focused onto a film or measured by a camera.
But TEM needs its electron beam to travel through a vacuum, which means placing the sample under vacuum conditions. Meanwhile, nanoparticles need to be in solution in order to assemble into nanocrystals. Placing a liquid sample in the TEM vacuum would cause the solution to evaporate, destroying the assembly process rather than capturing its image.
Researchers at Lawrence Berkeley National Laboratory solved the problem by sealing their solution in a liquid cell. The scientists were studying platinum-iron nanorods, which can act as catalysts in electrochemical reactions. This makes them good candidates for inclusion in devices that store energy and convert it from one form to another. The nanorods self-assemble from a “growth solution” of platinum and iron nanoparticles dissolved in a solvent.
To create a liquid cell that could protect the solution from evaporation in a TEM’s vacuum, the researchers stacked two thin transparent membranes of silicon nitride atop one other, leaving a gap only 120 nm thick between them. This gap was the right size to allow capillary action to suck the growth solution into the cell, leaving the solution sandwiched between the membranes. Finally, epoxy sealed the sandwich edges shut to close up the cell.
Another study in the same issue of Science used a similar technique to image nanocrystals assembling, but they were observing how two iron oxyhydroxide nanoparticles can attach to each other after they rotate to align their crystal lattices.
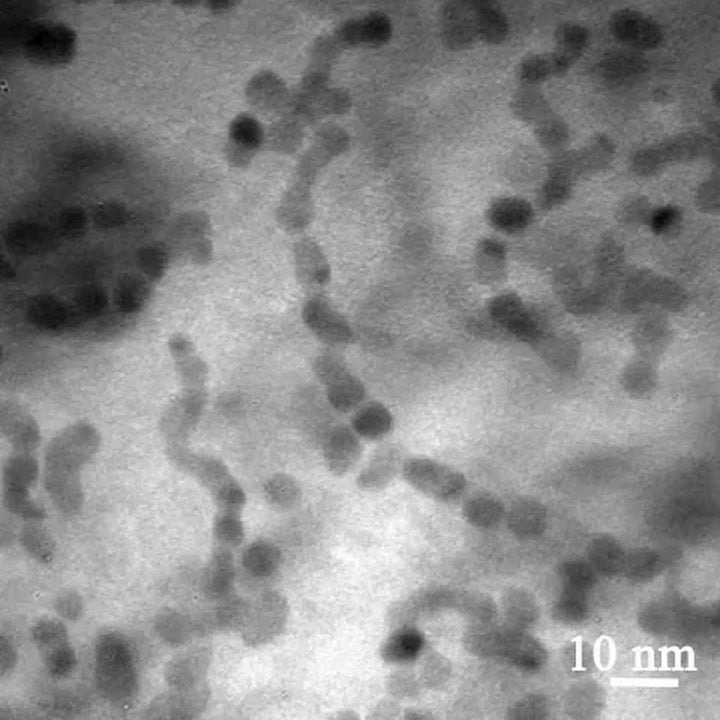
When the liquid cell of platinum-iron nanoparticles went into a TEM, this is what the scientists saw. These videos are sped up to run 30 times faster than real time. As time went on, the structures growing out of the solution became more and more organized. The nanoparticles began by forming long chains with multiple crystal orientations. Then the chains aligned, latching on to each other to form nanowires. Finally, the nanowires straightened out into long nanorods, 40 times longer than they were wide, with uniform crystal structures and orientations.
The paper describes in detail how the mess of nanoparticles grew more and more organized, eventually forming nanorods with a consistent crystalline structure:
“The nanoparticle chains formed during the early stage are winding and markedly flexible. The relative position of the nanoparticles within the chain changes and the orientation of individual nanoparticles also alters…During the final stage of growth, adjacent nanoparticles within the chain contact each other, forming a neck…Subsequently, mass redistribution eliminates the neck, and a smooth nanowire is formed. The diameter of the nanowire is slightly smaller (about 4.0 nm) than that of the individual nanoparticles before attachment (5.3 nm). A bent polycrystalline nanowire can straighten and transform into a singlecrystal nanorod with well-defined shape. This final stage of structural relaxation sometimes proceeds in parallel with the second stage of nanoparticle attachment. Notably, most of the nanowires remain twisted and polycrystalline for an extended period of time.”
Knowing how these rods form will help scientists design nanomaterials with specific properties for a more effective, and perhaps tinier, future.
Via Science