Researchers today unveiled a DNA nanorobot that can track down leukemia cells and kill them on sight, unleashing a therapeutic payload that causes the cancerous cells to self-destruct. Incredibly, this molecular assassin can accomplish this assignment while leaving healthy cells unharmed.
Leading researchers in the field of nanotechnology are calling the clam-like bots (which are assembled from the same components that make up your genetic code) a major milestone in the field of smart drugs. This level of praise has been hard-earned, and a long time coming; it’s taken the field of DNA nanotechnology thirty years to get where it is today. Find out how scientists got here, and how DNA nanotechnology — once considered by many to be a pipe dream — is now poised to change the future of medicine and technology forever.
What is DNA Nanotechnology?
Nanotechnologies are materials, structures, or devices intentionally designed by scientists to function on a scale of less than 100 nanometers. As a point of reference, a water molecule is about 1 nanometer across, while a single strand of hair has a diameter of about 100,000 nanometers. Researchers use a variety of molecules to create nanotechnologies (carbon nanotubes, for example, are popular in nanotech); but DNA nanotechnologists work with — you guessed it — DNA.
Why DNA? Compared to a lot of other microscopic building materials, the components that comprise DNA (and the way they interact with one another) are pretty well understood. Every strand of DNA is made up of a series of just four basic components, namely: adenine, thymine, guanine, and cytosine (abbreviated A, T, G and C). Collectively, these building blocks are called nucleotides. Bring two strands of DNA together, with nucleotides that complement one another in the right way (A with T, G with C), and they’ll pair up – or “hybridize” — with one another.
You’re all familiar with the double helix shown above; it’s the most commonly known conformation that two strands of DNA can take. But DNA strands can assume other forms, as well. For example, during a process known as genetic recombination, a total of four strands can join together in a transient configuration known as a Holliday junction.
One look at the Holliday junction — seen here — and you can begin to understand how DNA might be used to build more intricate structures than the double helix we all know and love. The thing about most naturally occurring DNA conformations like the Holliday junction, however, is that they are unstable — the strands only stay positioned in that particular conformation for a short while, and therefore make for pretty sorry building materials in nano-scale construction projects.
But scientists are clever. Remember the movie Gattaca? The name was a creative arrangement of the letters corresponding to DNA’s four nucleotide components; and much in the way that director Andrew Niccol arranged the letters A, T, G, and C to spell out the title of his film, scientists can join these individual building blocks together to create strands that spell out whatever they please. By piecing together custom-made strings of nucleotides, scientists can therefore create artificial DNA molecules capable of assembling into arrangements which — like the Holliday junction — have several “branches” of DNA, but are much, much more stable.
What Can You Build With This Stuff?
This figure illustrates the first (and also one of the most basic) examples of DNA nanoarchitecture — what Nadrian Seeman, the scientist who first conceived of DNA nanotechnology in the early eighties, calls “the fundamental notion” behind the entire field. On the left is a four-arm branched DNA molecule, with nucleotide tails (labeled X, X’, Y, and Y’) that can form structurally sound bonds with the tails from other, identical four-armed DNA molecules to give rise to the quadrilateral on the right.
“In the example shown,” explains Seeman, “only the inner [DNA strand ends] are used in forming the quadrilateral. Consequently, the structure can be extended to form an infinite lattice.”
It was 1982 when Seeman first demonstrated that junctions and lattices could be crafted out of DNA, but these structures were just the first in an ever-growing list of increasingly complex (and increasingly capable) DNA constructs. It wasn’t long, for instance, before researchers managed to design strands of nucleotides that could assemble into more intricate shapes — a cube, for example, or a tetrahedron. By the turn of the century, researchers had designed an actual machine in the form of “molecular tweezers” — a dynamic system of multiple DNA strands capable of opening and closing around other nano-scale objects.
Novel methods of DNA assembly were also developed and refined, most notably a technique called “DNA origami,” wherein a long strand of DNA is folded over onto itself and held together by smaller nucleotide strands (called staples). In 2006, the DNA origami method was implemented to great effect when researchers used the technique to craft a variety of intricate 2-D constructs, including a smiley face, and a map of the Western Hemisphere.
The Next Industrial Revolution
The first 3-D structure to be assembled via the origami method was created in 2009. A team of researchers from the Centre for DNA Nanotechnology in Denmark used the technique to create a self-assembling DNA box, designed to ferry around (then-theoretical) therapeutic cargo (the box, pictured here, even has a lid that can be opened with specially designed DNA keys); in 2010, researchers successfully operated the first assembly line comprised entirely of programmable DNA nanobots; and less than a year ago, researchers were predicting the imminent arrival of sophisticated, nano-scale drug-delivery vehicles capable of targeting diseased cells — which is precisely the type of nanorobot we see described in today’s issue of Science.
https://gizmodo.com/behold-the-first-nanobot-assembly-line-in-action-5538320
New Scientist’s Jessica Hamzelou summarizes the paper’s findings:
[A research team led by Harvard researcher Shawn Douglas] designed the device with DNA modeling software that understands how DNA base pairs bind together, as well as the helical structure that results. When they enter a shape of their choosing into the program, it returns a list of DNA strands that can be mixed together to create the desired shape.
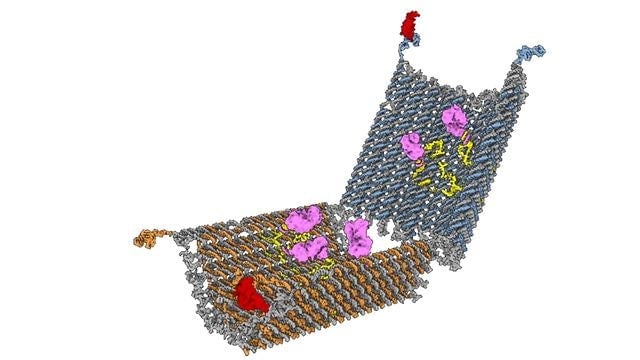
The shape that Douglas and his colleagues had in mind was clam-like, so that the nanorobot could hold a drug dose inside until it was time to deliver it. [The molecular structure of the team’s nanorobot is shown here]
To test its therapeutic potential, Douglas’s team created a nanorobot with locks that unzipped in response to molecules expressed on the surface of leukaemia cells. The team then loaded it with a single molecule known to kill cells by interfering with their growth cycle. Finally, they released millions of copies into a mixture of healthy and cancerous human blood cells.
Three days later, around half of the leukaemia cells had been destroyed, but no healthy cells had been harmed.
“The group provide proof of principle that DNA origami has the capacity to create highly intelligent drugs that activate only on encountering diseased cells,” explains Jørgen Kjems — who led the team from Denmark that created the first 3-D DNA origami back in 2009, but was not involved in the research. “This will inevitably lower the toxicity and side effects of the drugs carried within the device.”
Caltech’s Paul Rothemund, who used DNA origami to create the smiley face that we talked about earlier, echoed Kjems’ sentiments:
“Smart drugs which can be exquisitely targeted to specific cell types are a major goal of biomedical research,” he explained. “The ability to [match] the binding of the clam shell to the targeted cell type and use this as a trigger for delivery is a major step beyond the smart drugs of today.”
The next great industrial revolution has already begun, and it’s playing out on the microscopic level. In the last thirty years, DNA nanotechnology has grown from little more than a conceptual castle in the sky to a viable and promising means of computation and molecular architecture; and today, it’s paving the way for monumental advances in science, medicine, and industry.
In the same way that the last several decades have been marked by an evolution in electronics, the decades yet to come will be defined by an evolution in nano-scale biological machines.
The findings of Douglas and his team are published in today’s issue of Science.
Additional Resources
Read more about the Nanorobot unveiled today over on New Scientist
“Nanomaterials Based on DNA,” by Nadrian C. Seeman
“Nanotechnology and Cancer,” by James R. Heath and Mark E. Davis
DNA base-pairing structure diagram via; Holliday junction via Wikimedia Commons; DNA Lattice figure and 2-D DNA Origami via Seeman; 3-D DNA Origami via PhysicsWorld; Logic-gated nanorobot via Douglas et al.