Many people, myself included, have tried to lose weight at some point in our lives. And statistically speaking, the vast majority of the time, those attempts have ended in abject failure, with little sustained weight loss to speak of.
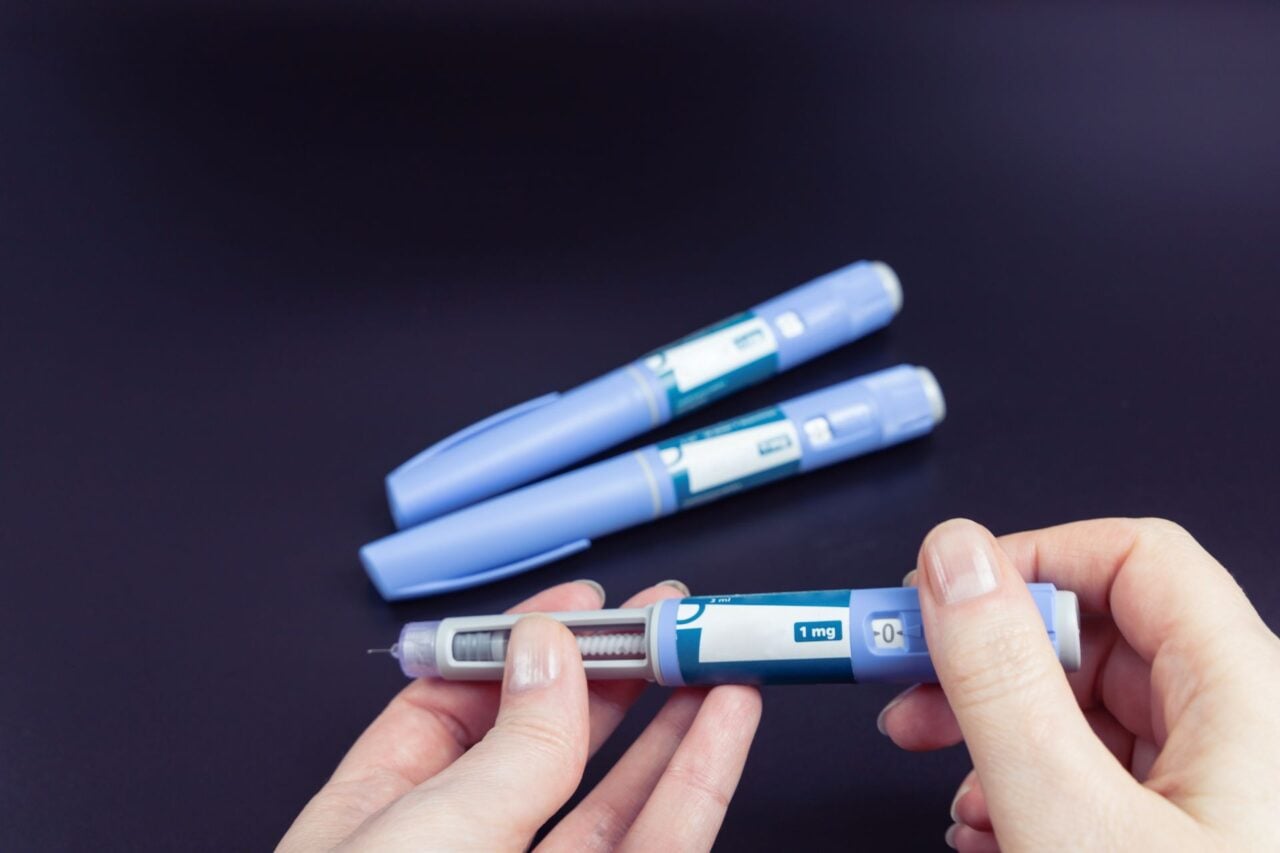
This grim outlook has started to change in recent years, thanks to the emergence of semaglutide, the active ingredient in Ozempic and Wegovy. Semaglutide mimics GLP-1, a hormone important to regulating our hunger and insulin production, among other things. The latest GLP-1 drugs (and newer iterations that target multiple hormones) have proven to be much more effective at helping people lose weight than diet and exercise alone, as well as basically any other intervention outside of the best bariatric surgeries.
GLP-1 drugs have their own limitations, including their very real side effects and the fact that many users will regain weight if they stop taking them. But they have truly ushered in a new era of obesity treatment, one that has finally started to see obesity rates in the U.S. and elsewhere decline.
In light of the upcoming 5-year anniversary of Wegovy’s approval, I figured it’d be fun to take a little stroll through the graveyard of past obesity treatments and discuss exactly why they failed.
2,4-Dinitrophenol (DNP)
2,4-Dinitrophenol, or DNP, has amassed a long resume over the past century. It’s been used in dyes, munitions, and herbicides, to name a few. In the 1930s, scientists discovered that it could also help people rapidly lose weight (up to three pounds a week). It does so by disturbing how our mitochondria use energy to produce ATP, our cells’ primary fuel, instead causing most of this energy to dissipate into heat. This then raises people’s metabolic rate and increases the burning of fats and carbs.
Unfortunately, people who take too much DNP can quite literally overheat themselves to death. Slightly more fortunate users can still experience severe side effects like cataracts, deafness, and kidney failure. Adding to the trouble is that DNP has a low therapeutic window, meaning there’s only a small difference between a “safe” and potentially deadly dose.
Countries like the U.S. quickly banned DNP as a weight loss drug once these side effects were apparent, but even today, it can be procured with relative ease. Plenty of people, particularly bodybuilders looking to gain an edge, are still gambling with its use, and some have unfortunately paid the ultimate price. On the more positive side, some scientists are still studying whether the mechanisms behind DNP can be safely exploited to treat obesity and related conditions.
Lap bands
Even in the GLP-1 era, several types of bariatric surgery are still highly effective at treating obesity, particularly for people with severe cases. While these surgeries are generally safe, though, they can be a costly and permanently life-altering treatment, which has limited their appeal. Laparoscopic gastric banding, better known simply as the lap band, was an attempt at addressing these concerns.
The minimally invasive and short procedure involves placing an inflatable, adjustable, and removable band around the top of the stomach. This effectively shrinks the size of the stomach, meaning it takes less food to feel full. Early research indicated that lap bands could provide reliable weight loss with minimal side effects. During the late 2000s, lap bands were briefly the most popular type of bariatric surgery.
Over time, however, it became apparent that people tended to experience more complications the longer they wore the bands, including infections, hernias, or the loosening of the device itself. As a result, many people regained their lost weight or at least required additional surgeries to repair their bands. Around the same time, doctors improved the safety and ease of now common surgeries like the sleeve gastrectomy.
Though the lap band was never formally banned, its popularity dropped like a stone by the late 2010s, and many bariatric centers today expressly do not even offer it.
Fen-phen
Fen-phen is a combination of the drugs fenfluramine and phentermine.
Both help suppress appetite, but work in different ways. They were originally approved as short-term obesity aids, with modest effects at best. In the late 1970s, however, pharmacologist Michael Weintraub started theorizing that mixing the two drugs could deliver better results than either could on their own. Fen-phen wasn’t widely used until the 1990s, but it rapidly became the weight loss treatment of choice for many. At the peak of its popularity, roughly 18 million prescriptions of it were doled out annually.
The drug combo was prescribed off-label, though, and had never undergone extensive testing in large clinical trials (unlike GLP-1s). Doctors, researchers, and patients soon began to report severe complications likely caused by fen-phen, including a higher risk of heart valve disease and pulmonary hypertension. By late 1997, the Food and Drug Administration pushed for fenfluramine and the related drug dexfenfluramine to be pulled from the market. This effectively ended the era of fen-phen for good, though not without its maker, American Home Products, losing billions in legal settlements.
The serious health risks associated with fen-phen were largely tied to the fenfluramine half of the combo. Phentermine is actually still prescribed occasionally for weight loss, often paired with the drug topiramate (this combo provides modest weight loss, but is generally safe). As for fenfluramine, it’s since gotten a second life as a treatment for certain kinds of seizures, though now with a clear warning of its potential cardiovascular risks.
Lorcaserin
At the time of its FDA approval in 2012, lorcaserin was noted for being the first novel medication approved for obesity in over a decade.
The drug was only ever modestly effective at helping people lose weight, though (perhaps up to six pounds more over a 12 week period). More damning was the realization that it likely raised people’s risk of cancer. A later FDA investigation of clinical trial data found that 7.7% of lorcaserin users developed cancer over a five year span, compared to 7.1% of people taking a placebo.
In 2020, the FDA requested that Eisai Inc, the makers of lorcaserin, remove the drug from the market, and the company complied. A year later, the FDA approved Wegovy. Funnily enough, some research has since suggested that GLP-1s might actually lower people’s overall risk of cancer.
Rainbow pills
Fen-phen was far from the first time that doctors tried to tackle weight loss through jamming together multiple drugs at once. The most flamboyant of these attempts were rainbow pills, nicknamed for the bright colors they were adorned in.
The heyday of the rainbow pill, at least in the U.S., was around the 1940s and 1950s. Pharmaceutical companies combined drugs thought to cause weight loss, including amphetamine stimulants, diuretics, and thyroid hormones, with drugs meant to counteract the well known side effects of the former, such as barbiturates to calm people’s anxiety or insomnia from the amphetamines.
Unsurprisingly, this logjam of drugs only served to make rainbow pills more dangerous, aided by the fact that people could be given widely varying doses from one pill to the next. At least sixty deaths in the U.S. were linked to rainbow pills before the FDA stepped in to ban such combinations by the late 1960s.
Rainbow pills still saw some popularity in other countries afterward, though. And some researchers have argued that many dietary supplements marketed today for weight loss are a repackaged version of rainbow pills. These products are far less regulated than approved drugs and they’re often mislabeled or even laced with some of the same types of drugs once common in rainbow pills, particularly stimulants.
Speaking of supplements…
Ephedra
Ephedra is technically the name for a genus of shrub plants, though its medical claims are typically associated with the stem and branches of the species Ephedra sinica. Ephedra’s primary ingredient is the stimulant ephedrine.
Starting in the 1990s, Ephedra saw widespread use as both a weight loss product and a potential performance enhancer for athletes. Often, it was paired with caffeine since some research had suggested the combination provided greater synergistic benefits. As the drug became more popular, though, reports of adverse events linked to it, including high blood pressure, stroke, and even psychosis. began popping up.
The final nail in the coffin for Ephedra, however, came when MLB baseball pitcher Steve Bechler suddenly died of heat stoke during spring training in 2003. An autopsy concluded that Bechler’s use of Ephedra contributed to his death. The FDA opened up an investigation following Belcher’s death, and about a year later, it took the rare step of completely banning Ephedra supplements containing ephedrine from the market. At least 155 deaths were linked to Ephedra prior to the ban.
Perhaps the bleakest footnote to this whole saga is that Ephedra wasn’t all that great in the first place. Even with caffeine, the drug only had modest weight loss benefits. Compared to placebo, it might have helped people lose one to two pounds more a month on average, according to a 2003 review.