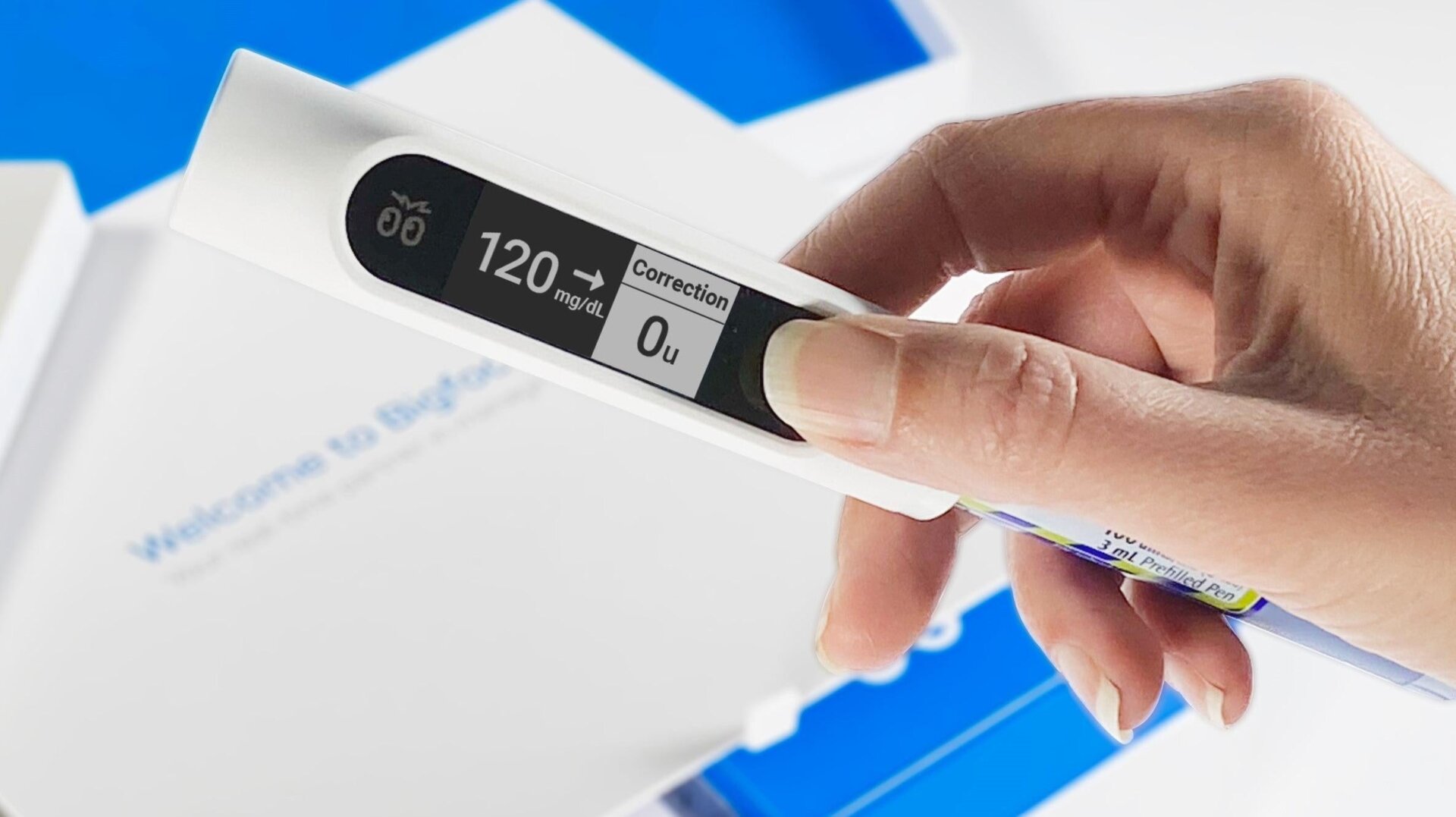
The Food and Drug Administration has granted approval for an innovative smart pen cap system to help those with both Type 1 and Type 2 diabetes better manage their insulin dosage. This is huge news, as the system could help diabetics eliminate the guesswork when it comes to figuring out how much insulin to take, using real-time data.
The Bigfoot Unity smart pen caps are made by Bigfoot Biomedical and are meant to be used by patients undergoing multiple daily injection therapy. The smart pen caps received 510 (k) clearance from the FDA, and according to the company, it’s the first system of its kind. It works in tandem with the Abbott FreeStyle Libre 2 integrated continuous glucose monitoring system (iCGM) to provide real-time blood glucose data. The user scans the sensor, and the cap then displays a patient’s blood sugar, a trend arrow, and a suggested dosage based on those values and what’s been recommended by their physician. Bigfoot Unity caps can be used by anyone ages 12 and up, and also work with all major insulin pens—both rapid and long-acting—from Eli Lilly and Company, Novo Nordisk, and Sanofi. (You can watch a video explainer of how the caps work here.)
As the caps also come with a companion app, users can also receive notifications when their blood sugar falls below 55 mg/dL, as well as reminders in the event they may have missed taking a long-acting insulin dose. The app also eliminates the need to enter these blood glucose values in a separate device. Instead, the smart pen caps automatically record the values and upload them to the cloud whenever it detects wifi or cellular signal.
“Diabetes management is incredibly hard because insulin has no fixed-dose or timing, leaving individuals to constantly determine their doses and configure devices as they make multiple critical decisions every day about how much insulin to take,” Jeffrey Brewer, CEO of Bigfoot Biomedical, said in a statement. “There are more than 7 million people in the U.S. on insulin therapy and many use insulin injections multiple times a day, yet there have been few advancements for this hugely underserved market.”
Blood glucose monitoring appears to be the next big thing in health tech. Both Apple and Samsung have been rumored to be working on non-invasive methods to measure blood sugar for the next-gen Samsung Galaxy Watch and Apple Watch. Another such blood glucose-monitoring smartwatch prototype was also showcased at this year’s CES. Meanwhile, in February, Fitbit launched blood glucose tracking within its app.
However, while it’s clear Big Tech definitely has its sights set on blood glucose, it’s hard to say when these advancements might be ready for consumer use. For starters, accurate blood sugar monitoring is non-negotiable. Right now, even ECG and abnormal heart rate notifications on smartwatches are treated as a tool for awareness. Any company that includes those kinds of features also must ensure its users understand that it’s not meant to be taken as medical advice or a diagnosis. Real-time blood glucose monitoring can have a much more serious impact on patients with diabetes—which means FDA clearance will likely be required. That in and of itself can be a complicated and lengthy process.
Given that context, it’s neat to see some FDA-cleared innovation to help streamline treatment in this area—and that there may not be too long of a long wait for those interested in trying it out. Bigfoot Biomedical says the smart pen cap system will be available through select diabetes clinics starting this spring.