America’s first plutonium implosion test—its first detonated nuclear bomb—left the 100-foot (30-meter) tower that held it aloft, including all its copper wire, copper sheathing, and coaxial cables, completely vaporized.
Almost instantaneously, the 21-kiloton fireball erupted into the New Mexico sky as Robert Oppenheimer initiated the July 16, 1945, Trinity test. The blast exposed the tower’s aerosolized metals and the sandstone desert dust beneath them to temperatures in excess of 2,732 degrees Fahrenheit (1,500 degrees Celsius). The pressure on these elements, roughly one million pounds per square inch, rivaled the pressures bearing down on rocks hundreds of miles underground where the Earth’s crust presses into its mantle.
Small wonder, then, that physicists, geologists, and other researchers are still finding radical reconfigurations of the desert Southwest’s basic chemical makeup over 80 years later in what was once the Manhattan Project’s Alamogordo Bombing Range.
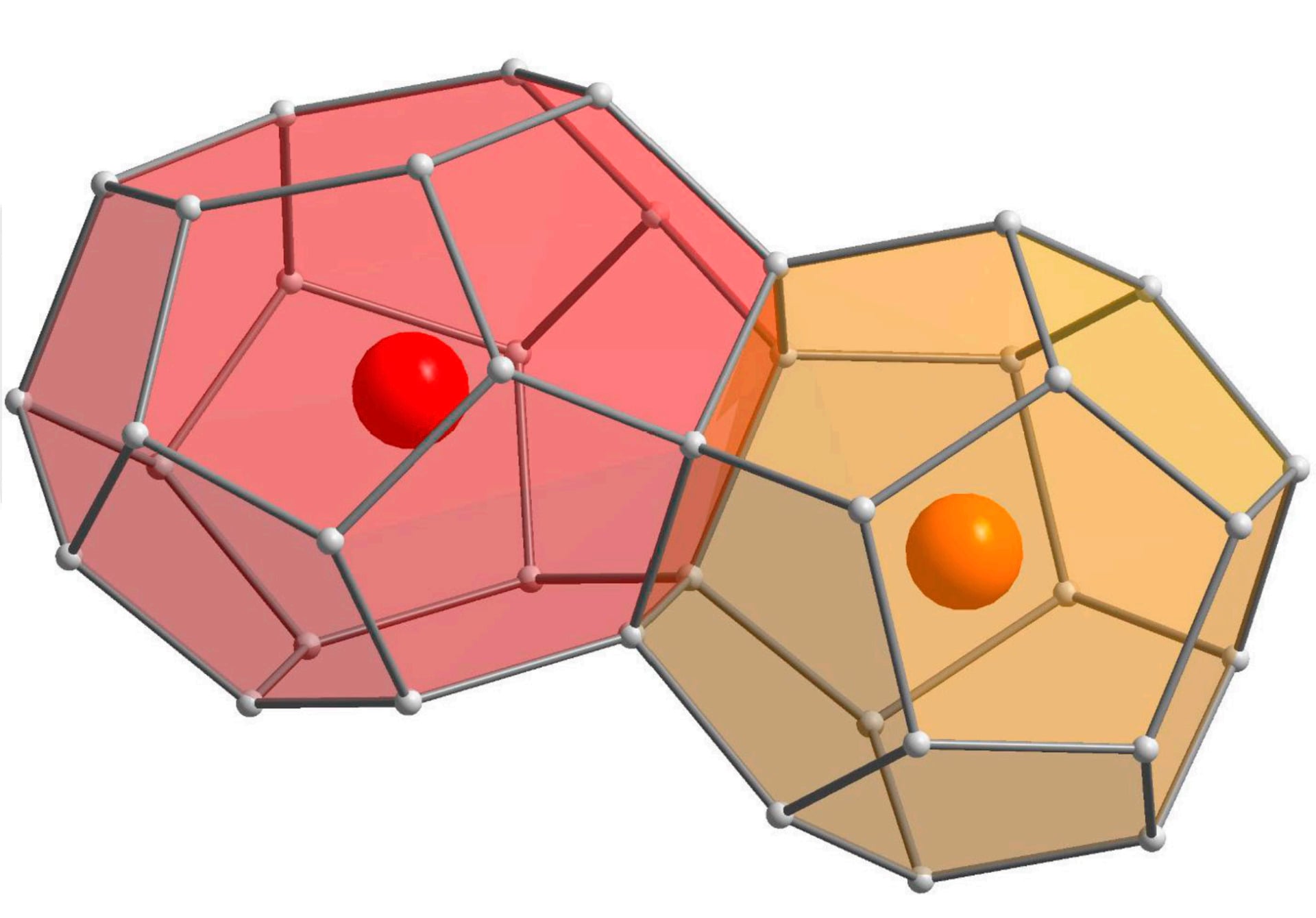
Now physicists in Europe and the United States have uncovered what they describe as a “previously unknown crystalline phase” of a type of compound, called a clathrate, composed of silicon, calcium, iron, and some of that copper evaporated by Trinity’s historic blast. Like other clathrates, this Trinity site crystal forms a complex geometric latticework capable of storing other smaller molecules or atoms like a nanoscale cage.
The discovery, as the new study’s lead author, Italian geologist Luca Bindi, and his colleagues wrote, “represents the first crystallographically confirmed identification of a clathrate structure among the solid-state products of a nuclear explosion.” The discovery could deepen our understanding of these valuable crystalline compounds, the researchers said.
Crystal chemistry
Material scientists prize clathrates for their utility in myriad high-tech applications. These crystals can, for example, act like little parking garages storing the lithium ions within lithium-ion batteries, both at the beginning and end of charging and discharging cycles. And, across industries, clathrates have helped engineers tailor highly bespoke silicon compounds, implanted or “doped” with elements whose specific electrical, magnetic, or catalytic properties can enhance solar cells, quantum computers, and countless other innovative devices.
“More broadly,” as Bindi and his coauthors put it in their new study, “this work underscores how rare, high-energy events—such as nuclear detonations, lightning strikes, and hypervelocity impacts—serve as natural laboratories for producing unexpected crystalline matter.”
The newly discovered clathrate crystal will also help scientists better model and predict how these complex molecular geometries form by serving as a truly nuclear edge case, one “beyond the reach of conventional synthesis,” they wrote in the study.
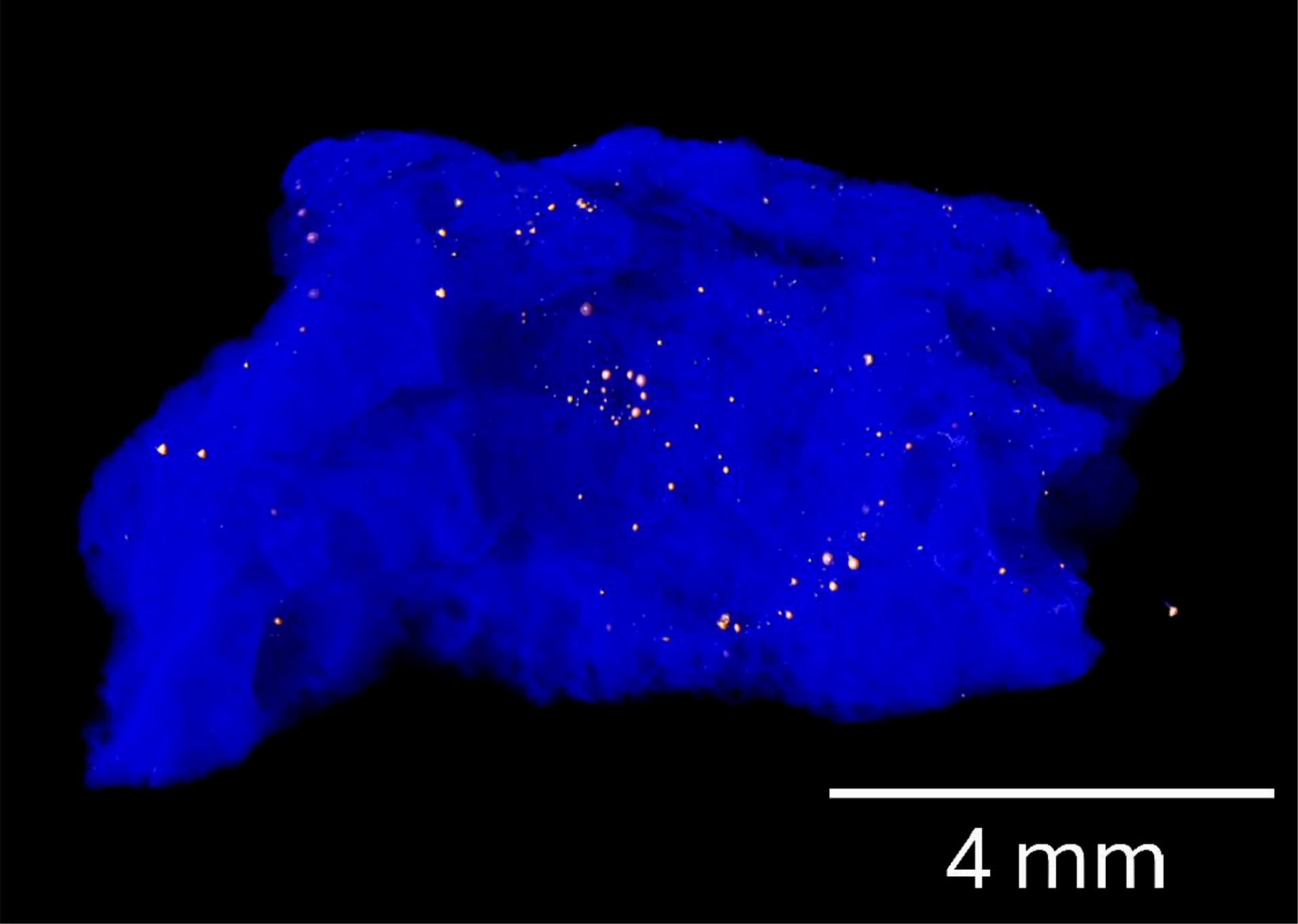
Crystal keys
The researchers found this previously unknown clathrate inside red trinitite, a rare fragment of glassed sand produced by the Trinity bomb.
“While the majority of trinitite exhibits a characteristic pale green color,” the researchers note, “a less common variety—red trinitite—is enriched in metals derived from the vaporized tower, coaxial cables, and recording instruments.”
Bindi, the chair of mineralogy and crystallography at the University of Florence in Italy, worked with physicists at Princeton, Carnegie Mellon, and the Slovak Academy of Sciences to subject this clathrate material to highly detailed single-crystal X-ray diffraction analysis to map its 3D geometry. They then experimented with the clathrates’ quite literally multifaceted geometric structure—its dodecahedral (12-faced) and tetrakaidecahedral (14-faced) silicon cages—to determine whether it could be related to another unusual quasicrystal they found at the Trinity site in 2021.
While the researchers could not determine a relationship between these two unusual compounds forged by the nuclear blast, they noted that many unusual and still undiscovered compounds exist inside the site’s red trinitite, waiting to be compared and contrasted.
“Systematic investigation of metallic droplets in red trinitite has revealed a range of unusual phases,” Bindi and his coauthors noted in the study, “reflecting the unique chemical environments produced during the explosion.”






